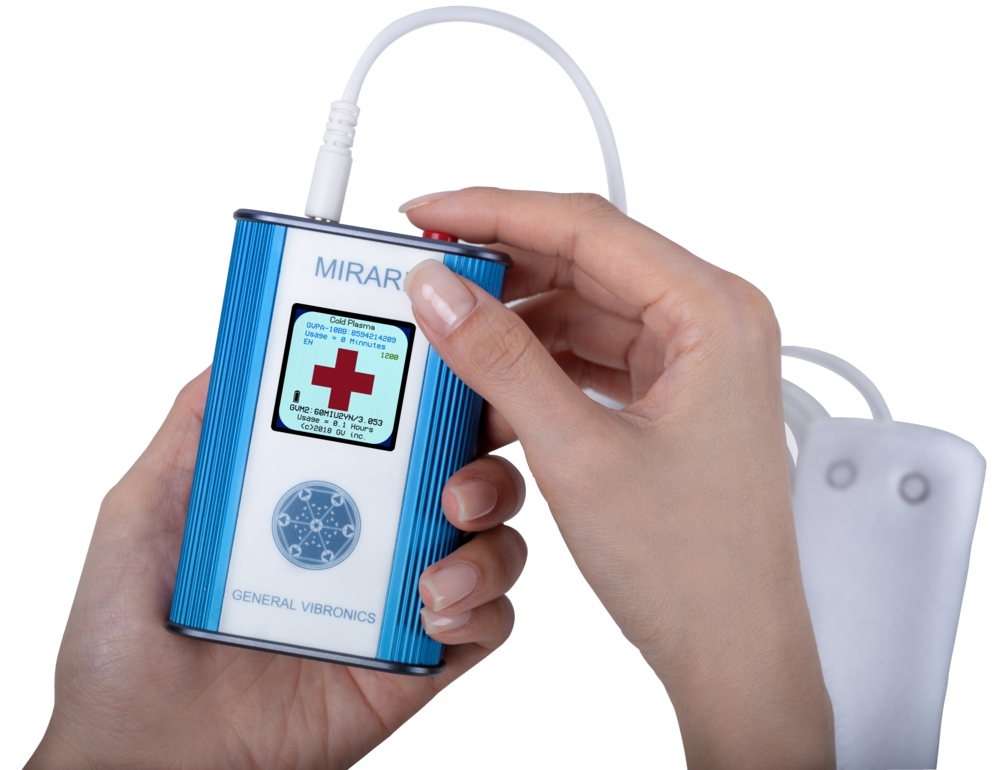
Pioneering RF energy, Dielectric Barrier Discharge (DBD) technology for modern medicine
|
Mirari Doctor Pte. Ltd. is a Singapore-based medical technology company bringing FDA 510(k)-cleared therapeutic solutions to clinics worldwide. Our flagship offering, the MIRARI® Cold Plasma System — developed by General Vibronics, Inc. — harnesses Dielectric Barrier Discharge (DBD) technology at 80 kHz to deliver targeted radiofrequency energy for temporary relief of pain, muscle spasms, and improved local circulation.

The MIRARI® Cold Plasma System designed and manufactured by General Vibronics, received U.S. Food and Drug Administration (FDA) cleared in November 2024 (Certificate No.3959-1-2025-1) marking a significant milestone in its validation for medical use.